By Allen P. Green, M.D. May 2026
I spent today at BerkeleyCAL, the Berkeley Conference on Aging & Longevity, at UC Berkeley’s Stanley Hall, where Dr. Michael Conboy presented work from his and Dr. Irina Conboy’s lab that deserves to be flagged for anyone following the science behind therapeutic plasma exchange. The first author is Joana Marie C. Cruz; the paper was just accepted in Aging Cell and posted online this past week. Dr. Conboy’s talk title was as direct as it gets: Plasma dilution after a heart attack helps heart recovery, repair and function, in mice.
The results are remarkable. But for those of us who have followed the Conboy lab’s earlier work, particularly their 2020 paper showing that a single plasma dilution rejuvenated tissues across all three germ layers in old mice, they are not entirely surprising. The Cruz paper extends that paradigm into the cardiovascular system, and does so under conditions that map onto the most common reason older adults end up in a cardiac unit: a heart attack.
Dr. Dobri Kiprov, our practice’s medical director, is presenting tomorrow at the same conference on parabiosis and human longevity, a fitting bookend to a meeting where the plasma-dilution story has now expanded from tissue rejuvenation into acute cardiac injury and recovery.
What the Study Did
The investigators used 18- to 22-month-old C57BL/6 mice, biologically equivalent to humans over 60, which is the age group where myocardial infarction risk climbs sharply. Mice were assigned to four groups: sham surgery, sham plus neutral blood exchange (NBE), ischemia–reperfusion (I/R) injury alone, or I/R plus NBE. The I/R model induces a controlled heart attack by ligating the left anterior descending coronary artery for 30 minutes, then restoring blood flow.
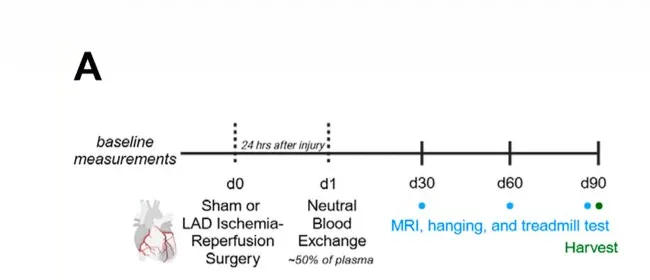 Study design showing the 24-hour delay between cardiac injury and plasma dilution, followed by monthly functional assessments for three months. Adapted from Cruz et al., Aging Cell 2026, CC BY 4.0.
Study design showing the 24-hour delay between cardiac injury and plasma dilution, followed by monthly functional assessments for three months. Adapted from Cruz et al., Aging Cell 2026, CC BY 4.0.
What is clinically interesting is the timing of the intervention. After a heart attack in humans, the immediate priority is restoring blood flow: catheterization and stent placement as fast as possible. Nothing about this study changes that. What the Cruz paper asks is a different question: once the acute intervention is done, is there something worth doing systemically in the hours that follow? NBE was administered 24 hours after the I/R injury, after the initial ischemic insult and after reperfusion, but before the peak inflammatory and fibrotic response that unfolds over the following 72 to 96 hours. In mice, the answer was yes.
The procedure replaced approximately 50% of each animal’s plasma with saline containing 5% albumin, the small-animal analog of what therapeutic plasma exchange does in human patients. Animals were then followed for three months, with cardiac MRI, treadmill testing, and grip-strength assessments at 30, 60, and 90 days, plus tissue analysis at 5 days (early molecular response) and 90 days (chronic remodeling).
What They Found
Cardiac structure and function. On serial cine-MRI, mice that received I/R plus NBE preserved their left ventricular volumes and ejection fraction; values were statistically indistinguishable from sham animals. I/R-alone mice developed the expected progressive ventricular dilatation and decline in ejection fraction over three months. NBE, in other words, prevented the post-infarct remodeling cascade that defines heart failure.
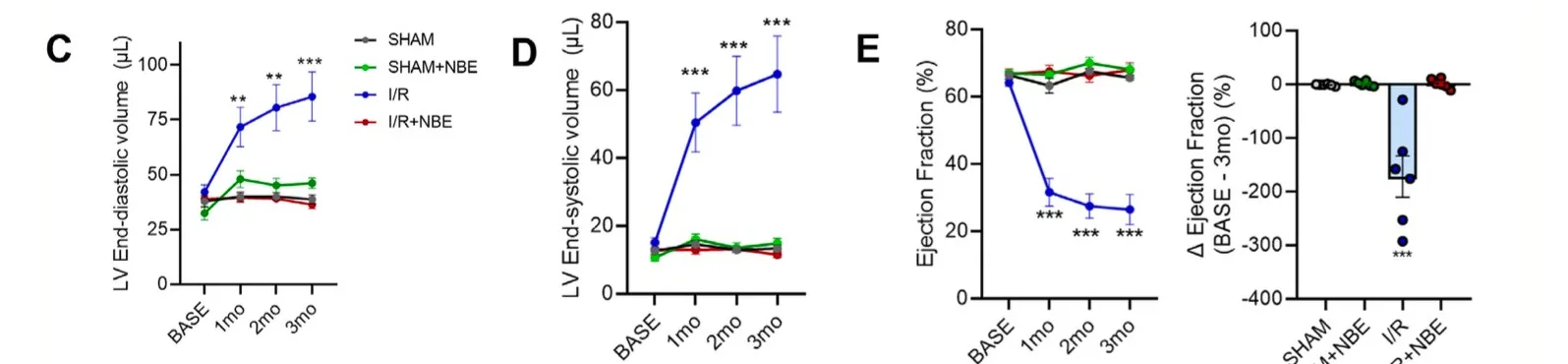 Monthly cardiac MRI measurements over three months. In every measure (end-diastolic volume, end-systolic volume, and ejection fraction), the I/R group (blue) diverges sharply while the I/R+NBE group (red) tracks with sham controls. Adapted from Cruz et al., Aging Cell 2026, CC BY 4.0.
Monthly cardiac MRI measurements over three months. In every measure (end-diastolic volume, end-systolic volume, and ejection fraction), the I/R group (blue) diverges sharply while the I/R+NBE group (red) tracks with sham controls. Adapted from Cruz et al., Aging Cell 2026, CC BY 4.0.
Physical performance. Hanging time (a grip-strength measure) and treadmill running distance both dropped by more than 50% from baseline in I/R animals over three months. In the I/R plus NBE group, both metrics remained comparable to sham. Notably, sham animals that received NBE (old mice with no heart attack, just the plasma dilution) outperformed sham controls on both measures. That finding is worth pausing on. It means plasma dilution improved physical function in aged animals independent of any disease or injury, consistent with the broader rejuvenation literature and directly relevant to the preventative medicine use case that brings most patients through our doors.
